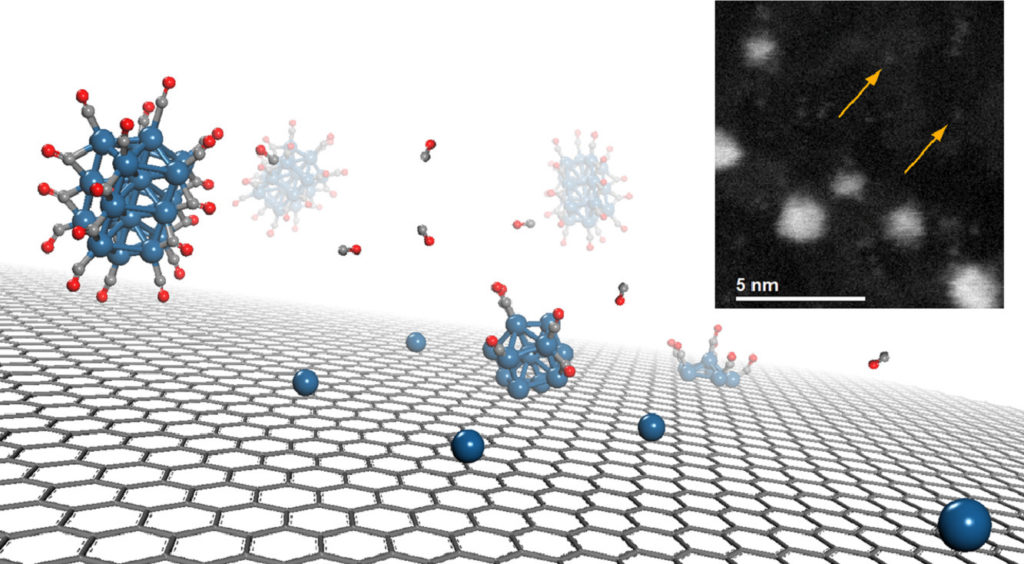
Having single atoms or small clusters docked onto a single layer graphene represents a charming feature for energy-storage and catalysis. Unfortunately, the large cohesion energy of transition metals often prevents the isolation of nanoscopic clusters, which invariably tend to aggregate. The decoration of defective graphene layers with single Pt atoms and sub-nanometric clusters is herein achieved by exploiting metal carbonyl clusters, as precursor, and investigated by means of transmission electron microscopy and X-ray photoemission spectroscopy. Unexpectedly, the process of aggregation of Pt into larger clusters is inhibited onto the surface of defective graphene, where the Pt-clusters are found to fragment even into single metal atoms.
Reproduced with permission. Copyright 2019, Elsevier